

GMP (Good Manufacturing Practices) Certification ensures that products are consistently produced and controlled according to quality standards. It is essential for manufacturers in pharmaceutical, food, cosmetics, and healthcare industries to maintain safety and compliance.

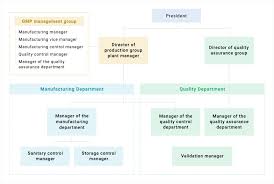
To obtain GMP (Good Manufacturing Practice) Certification, the following documents and details are required for compliance and audit purposes.
Certificate of Incorporation, Partnership Deed, or MSME Registration.
Valid manufacturing license issued by the concerned authority.
Complete list of products along with composition and labeling details.
Layout plan of manufacturing unit showing production and storage areas.
List of machinery and equipment used in manufacturing process.
Standard Operating Procedures and quality control documentation.
Good Manufacturing Practice Compliance